|
This was the first at-home diagnostic test using saliva samples, and it originally could only be obtained with a prescription from a doctor. It wasn't until May 8 that the FDA authorized Rutgers Clinical Genomics Laboratory to actually start testing samples of saliva collected at home for the coronavirus, using collection kits from Spectrum Solutions. On April 13, the FDA granted Rutgers University's RUCDR Infinite Biologics biorepository emergency authorization for a new method it developed to test saliva samples for the coronavirus. These tests are't invasive and can easily be done at home, which helps protect health care professionals from being exposed to the coronavirus at testing sites, and reduces the demand for personal protective equipment. 
Much like taking a DNA test, saliva tests have you spit into a tube, seal it and send to a lab. The Spectrum Solutions saliva collection device used for at-home saliva tests. Once people are aware that they are infected, they can take the proper measures to avoid others to prevent exposure. More than half a year into the pandemic, testing remains a crucial element of slowing the spread, because it identifies people who are carrying the virus, both those who are ill and those who aren't exhibiting any symptoms. These tests - of which there are now eight authorized for use - eliminate the need for someone who is feeling sick to go to a testing site or clinic, which in turn means that fewer people are exposed to the virus. The US isn't struggling as much now with a shortage of coronavirus test kits and related supplies, thanks partly to the advent of at-home testing. 
You needed a doctor's orders to get tested, and to get those orders, you had to meet specific criteria, such as having a fever and showing other COVID-19 symptoms. Consumers can access the Testing Locator at the most up-to-date news and information about the coronavirus pandemic, visit the WHO and CDC websites.ĭuring the first few months of the coronavirus pandemic, COVID-19 tests were in extremely short supply. People without health insurance do not have to pay for COVID-19 testing at ICATT locations. The tests are billed to third-party payers, such as Medicare, Medicaid, and private health insurers. 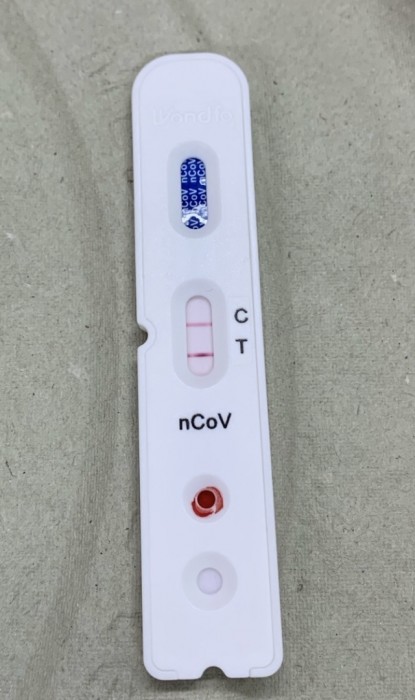 
Testing is available at pharmacies, commercial laboratory sites, community sites, and retail locations.ĬOVID-19 testing is available at no-cost at ICATT sites to people with or without health insurance who are experiencing symptoms or have been exposed to someone with COVID-19. Results are typically provided within 24–48 hours. Tests offered may include laboratory-based nucleic acid amplification tests (NAATs), including polymerase chain reaction (PCR) tests, and rapid antigen point-of-care (POC) testing. The locator is part of the CDC Increasing Community Access to Testing (ICATT) program, which provides access to COVID-19 testing, focusing on communities at a greater risk of being impacted by the pandemic, people who do not have health insurance, and surge testing in state and local jurisdictions. Today CDC launched the COVID-19 Testing Locator website, which will allow consumers to search for free COVID-19 testing sites near them.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
